Ever wondered why some things are sticky and others aren’t? Applied mathematician SOPHIE CALABRETTO explains the chemical attraction of garlic and the magic of tackifiers.
Apply knowledge of elements, compounds and chemical reactions, and learn about chemical attraction in this resource about the chemistry of stickiness and the mechanisms that control it. This resources is most suited to Year 7, 8, 9 and 10 students studying chemical and biological sciences. Download the student activity and teacher guide (including full lesson plan) to design your own sticky tape tester!
Word Count: approx.1074

As someone who loves all things fluid, it seems strange that it’s taken me until this year to wonder about stickiness and the mechanisms that control it. It seems like a trite observation, but lots of things are sticky – foods, plants, the eponymous tape. It’s been something I’ve taken for granted.
But is all stickiness the same? And are any solids truly sticky, or does the stickiness result from a sticky surface fluid?
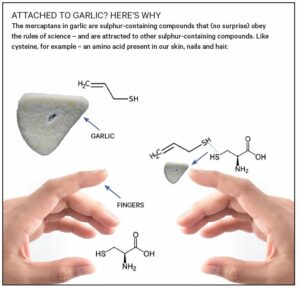
My inspiration came as I was chopping fresh garlic. It turns out that the stickiness I felt comes from mercaptans, sometimes lovingly referred to as “thiols”. These sulphur-containing compounds are attracted to other sulphur-containing compounds such as the amino acid cysteine, which can be found in our hair, nails and skin.
When those fresh garlic mercaptans come into contact with the cysteine in your skin, they form a strong bond. Think of this intermolecular bond as you would magnetism: you can’t really “see” the bond, but you can feel it.
This chemical attraction is what creates the physical sensation of stickiness on your fingers, and the stronger the bond, the stickier it will feel.
After gin, my second favourite substance is sugar which, by itself, is not sticky. White sugar, in its crystal form, is made of carbon, hydrogen and oxygen atoms, and when these relatively large molecules are dry and intact, they’re pretty impervious to each other, thanks to those strong internal oxygen-hydrogen bonds in their crystalline lattice. But with the addition of water, everything gets more complicated. The oxygen atoms around the outside of the sugar molecules have a slight negative charge; when dissolved in liquid, the sugar molecules disperse and those oxygen atoms are attracted to the hydrogen atoms in the water, which have a slight positive charge, because opposites attract – except on dating apps.
This chemical attraction is an example of hydrogen bonding and, although it’s not the strongest of the chemical bonds, it’s still enough for the sugary liquid to feel sticky.
“But my hands get all sticky when I touch fairy floss, and that’s dry!” I hear you proclaim. While it’s true that fairy floss is essentially many, many thin strands of molten sugar that have re-solidified as they’re spun up into a delicious mass, it sticks to you because your skin is covered in about three million sweat glands, and the average person sweats up to 5.6L per day. (Even if you’re not moving around that much, you’re looking at over 3L of daily sweat.) We’re all sweaty, all the time, almost everywhere. Unfortunate, I know.
Bio-sticky
Stickiness gets quite dramatic in the plant world. I love the excitement of a carnivorous plant trapping its prey – and many of these flesh-eating botanical marvels, such as sundews, are excellent stickymakers.

Sundews produce glistening drops of sticky, sweet mucilage (yes, a lot like mucus) to attract and entrap their prey. Their prey either dies from exhaustion trying to escape, or asphyxiation as the mucilage envelops them (gross). This bio-adhesive is a polysaccharide substance, containing long chains of monosaccharides – aka simple sugars. We know what happens when we have a whole lot of sugar plus moisture … stickiness!
But do not worry, dear reader – not all mucilage is used for evil. That delightful gooey substance inside aloe vera that you may rub on your sunburn is also mucilage. Some plants are good people.
Glue trap
Of course, we can’t talk about stickiness without referring to glue and adhesives: substances with the sole purpose of bonding things together. Adhesives come with a whole gamut of sticking methods, including drying (solvent-based and polymer dispersion adhesives), pressure (pressure-sensitive adhesives), contact adhesives, hot-melt adhesives and numerous others. But their sticky feeling is just a manifestation of their bonding process.
I’m less interested in how to accidentally glue my fingers together than I am in things that feel sticky when I touch them, so let’s talk about the kinds of adhesives that release their grip when I stick them to myself, like duct tape and Post-its. These are called “soft adhesives” and are a type of pressure-sensitive adhesive (PSA).
PSAs are usually composed of an elastomer – which is a polymer with viscoelasticity – often paired with a delightfully named “tackifier”, which is not only a compound used to increase the tack or stickiness of an adhesive’s surface but also my new favourite word.
Viscoelasticity means that, when deformed, a material exhibits properties of both a viscous fluid – thick molasses, for example – and an elastic solid, like a rubber band. Viscosity describes a fluid’s resistance to movement (the higher the viscosity, the larger the force needed to make it flow), and elasticity describes a solid’s resistance to deformation – for example, how much force you need to stretch that rubber band.
Viscoelastic materials can have elements of both of these properties, depending on the magnitude and the timescale of the stress being applied. This is very important for soft adhesives, because there needs to be a balance between flow and resistance for bonds to form and create adhesion.
Unstuck?
There are two very distinct ways in which they can unstick: either exactly like a solid or exactly like a fluid, with absolutely no crossover between them.

When you unstick a solid, a very thin slice of air appears between the adhesive solid and the adhering surface, breaking the bonds between the molecules in a way that does not deform the solid. Think of when you rip that picture hook off the wall – the adhesive strip doesn’t deform, and pulls away the layer of paint, which is more strongly bonded to the adhesive than to the layer of paint below.
Unsticking a liquid, on the other hand, sees very extensive deformation of the liquid adhesive, and air enters and breaks the bonds in a Saffman-Taylor-like behaviour (which creates a finger-like pattern in liquids with different levels of viscousness). This kind of unsticking is like removing your picture hook by pulling the tab at the bottom so the tape deforms and comes away neatly.
It turns out that stickiness is more amazing than I thought – and it all comes down to the physical effects of chemical bonding. Speaking as a mathematician, I was going to make a chemistry joke – but I’m afraid I won’t get a good reaction.
This article was written by Sophie Calabretto for Cosmos Magazine Issue 94.
SOPHIE CALABRETTO is a mathematician specialising in fluid mechanics, and Honorary Associate Professor at the ACE Research Group, University of Leicester. Her story on sharks appeared in Issue 89.
Cosmos magazine is Australia’s only dedicated print science publication. Subscribe here to get your quarterly fill of the best Science of Everything, from the chemistry of fireworks to cutting-edge Australian innovation.
Login or Sign up for FREE to download the educational resources





