Messenger RNA technology, so long in the wings, has leapt from understudy to main act. Its deceptively simple technology has potential well beyond our current COVID predicament.
This topical article could be used with students in years 8 to 12 studying Biological and Chemical sciences.
Word Count: 1622
[PLEASE NOTE: The information in this article may since have been updated or refined.]

In 1989, when Katalin Karikó submitted her first grant application to develop messenger RNA-based gene therapy, she knew she had a game-changing technology in her hands. But the Hungarian biochemist, then a professor at the University of Pennsylvania, could only dream of using mRNA in humans.
In theory an almost perfect platform to make drugs and vaccines, mRNA was stacked with practical problems that would keep it away from clinical use for decades. Then, at the end of 2019, a strange new infectious disease appeared in Wuhan and spread quickly across the planet, changing the world and sending countries and companies racing for a medical treatment that would stop the pandemic’s illness and fatalities.
mRNA has become one of the saviour technologies of the COVID era; now, that success is just the first step in a transformation in how we might recognise – and cure – everything from the flu to cancer.
The concept behind mRNA technology is strikingly simple. mRNA – which stands for messenger ribonucleic acid – is a single-stranded chain of nucleotides. In an organism, it acts as a messenger, a short-lived intermediary that communicates the information contained in our genes to the ribosomes. These are the cell’s protein factories, which read the code and translate it into a protein.
Special delivery: how mRNA technology works at a cellular level

1. Uptake by endocytosis
The liposome, or nanoparticle, is swallowed by the cell through endocytosis, the process by which cells absorb external material by engulfing it in a pouch – vacuole – of cell membrane called an endosome.
2. Endosomal escape
The endosome’s membrane breaks down, and the liposome is released inside the cell.
3. Release from carrier
The presence of enzymes inside the cell degrades the liposome. The mRNA strings are released and can travel to the protein-making ribosomes.
4. Translation
In the ribosomes, the mRNA strings are read and translated into antigens – pathogen proteins. The resulting antigens are exposed to the cell surface, where immune system cells recognise them as foreign, triggering an immune response that creates a memory for the antigen. If the real pathogen appears, immune cells recognise the same antigen and attack.
Scientists have learned to transcribe a genetic sequence from a string of DNA to a string of mRNA. A synthetic mRNA sequence with the right blueprint can be turned into a drug that, like a message in a bottle, delivers instructions into a cell to turn it into a literal bodyguard through its specialised resulting protein.
In a vaccine, the mRNA string encodes the recipe to make the antigen – a protein from the pathogen we want to protect ourselves from. Once the vaccine is inside the cells, the instructions are used to synthesise the antigen, which is exposed to the cell surface. Then, a subset of immune system cells recognises the antigen as foreign, triggering an immune response. This mechanism creates a memory for this antigen. Later, when the real pathogen is present, those cells recognise the same antigen and react rapidly and strongly against the infectious agent.
After the protein has been produced, the mRNA is degraded via physiological, metabolic pathways. “It’s a transient thing,” says Associate Professor Archa Fox, a molecular biologist and mRNA expert at the University of Western Australia. This desirable trait reduces the risk of unwanted side-effects by uncontrolled protein expression.
Two main issues had hindered the advance of the technology. “One was what we call a delivery problem,” says Fox. That same transience that makes mRNA desirable is also a problem: how to protect it from degradation during its journey throughout the body and into cells. In the late 1990s, researchers were able to pack the fragile messenger into shells of fat molecules called lipid nanoparticles, which had been studied for almost two decades as a possible delivery mechanism for anti-cancer drugs. The lipidic vehicle protects mRNA from thermal degradation and shields it from destructive enzymes while shunting it to the cell.
There was another major problem. The body strongly rejects RNA from outside sources, probably to avoid being hijacked by pathogens, and in early studies the mRNA often proved so toxic that it killed the lab animals it was tested on.
It took until 2005 for Karikó, who now oversees mRNA research at BioNTech, to discover that by adding pseudouridine into the mRNA she could fool the cell into thinking that the delivered mRNA was not a foreign invader. This breakthrough laid the foundations for the apparent overnight success of today’s mRNA COVID-19 vaccines.
For over three decades, researchers across the world had been working on mRNA therapeutics that can instruct the body to make its own drugs. With COVID, that platform has reached industrial scale, and offers opportunities for cures that have eluded answers thus far.
“It’s a flexible platform,” says Professor Thomas Preiss, a molecular biologist and mRNA expert at the Australian National University.
Custom cancer vaccines
Perhaps the holy grail of medical science in the 21st century is a cure for cancer. In recent years, cancer vaccines and immunotherapies have promised an alternative to treating malignancies.
Preventive cancer vaccines have been approved for some cervical cancers, head and neck cancer, and liver cancer. Therapeutic cancer vaccines are clinically used for early-stage bladder cancer and prostate cancer.
To every patient their own cancer vaccine
Cancer vaccines could revolutionise treatment by encoding the recipe for cancer-specific antigens of each patient. The vaccine “wakes up” the patient’s immune system to recognise cancer cells and kills them.
1. Cancer analysis
The DNA from a single patient’s cancer cells is genomically sequenced to identify antigens specific to that patient’s cancer. The antigens’ genetic codes are transcribed into an mRNA string.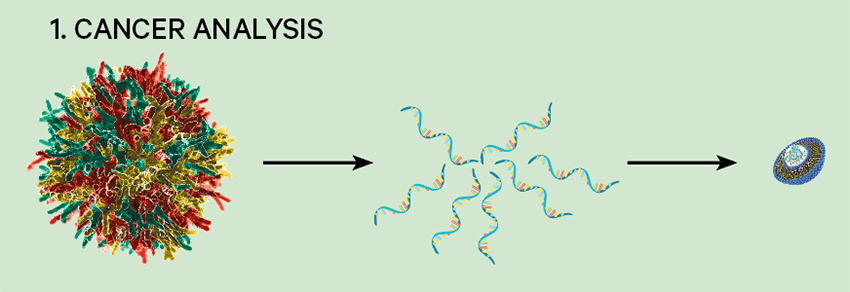
2. Vaccine delivery
A personalised vaccine is injected into the patient, whose cells are then capable of flagging the cancer’s antigen protein on their surface and triggering an immune response.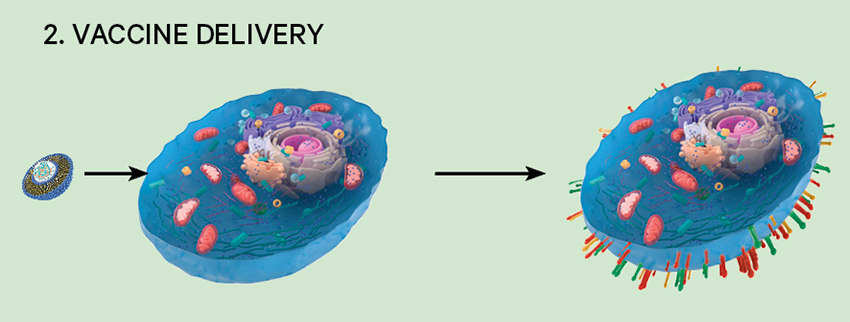
3. Immune activation
T-cells – killer cells that are part of the immune system – recognise the antigen protein as foreign and memorise it for immediate and future attacks.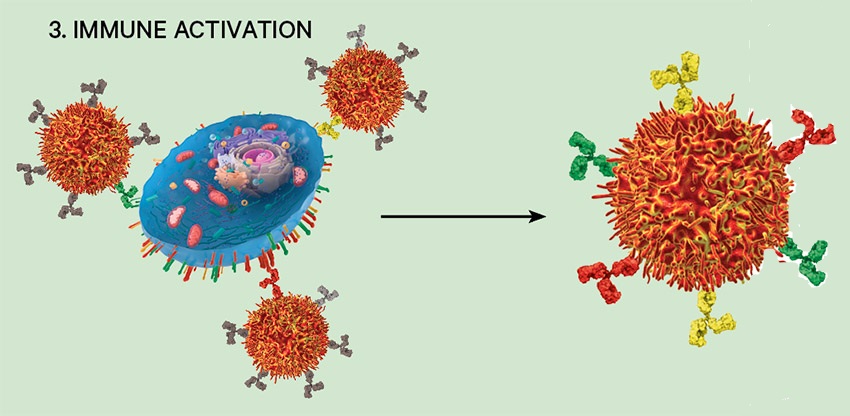
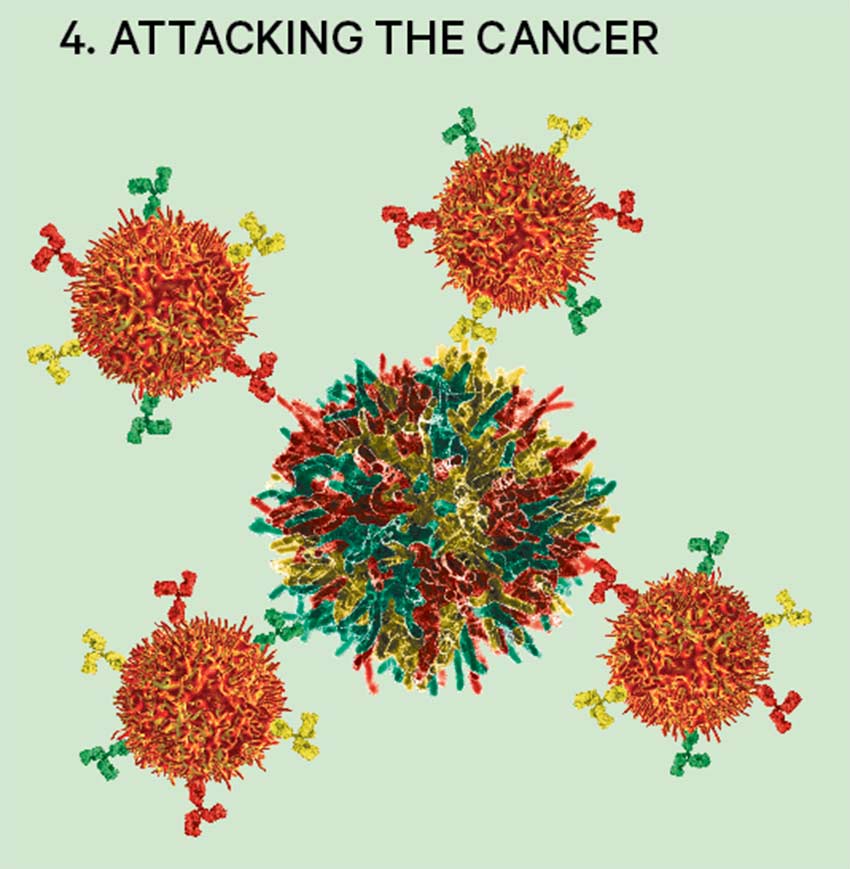
4. Attacking the cancer
T-cells can now identify cancer cells as invaders and latch on to them, causing their apoptosis – cell death – without affecting other cells in the body.
Illustrations: Greg Barton. Elements: Fancy Tapis, Ttsz, Sciepro / Getty Images
Researchers at Pfizer, Moderna and a large number of other biotech companies are exploring mRNA vaccines in which the mRNA containing the recipe for cancer-specific antigens is delivered into T-cells – killer cells that are part of the immune system. These are instructed to recognise cancer cells and causes apoptosis – cell death – without affecting other cells in the body.
“Traditionally, therapeutics have targeted the tumour, but now we target the immune system,” says Professor Peter Leedman, a cancer medicine researcher at the Harry Perkins Institute in Perth. Because cancer cells differ widely between patients, the magic of the mRNA vaccine is its ability to be tailored to fit the antigen repertoire of each patient.
The theory is simple. Scientists analyse the DNA from the patient’s cancer cells to identify antigens specific to that patient’s cancer. They transcribe the antigen’s genetic code into an mRNA string and inject it back to the patient’s T-cells to direct the immune system’s attack against the cancer cells.
These personalised cancer vaccines are designed to deliver one custom-tailored therapy for one patient at a time.
It’s a revolution that could transform cancer treatment. Although chemotherapy and radiotherapy have become more and more tailored over recent decades, they’re still the equivalent of a sledgehammer compared to the potential fine artist’s chisel of personalised mRNA therapy. A fair comparison might be the design and manufacture of a prosthetic limb: a tailored, one-off piece of technology intended to give its recipient the maximum opportunity to live as “normally” as possible. It is, without exaggeration, life-changing.
The first proof-of-concept studies that proposed the idea of mRNA cancer vaccines and provided evidence of the feasibility of this approach were published more than two decades ago. Today, several cancer vaccine candidates are undergoing pre-clinical and clinical trials around the world.
A bright future
Scientists have long known the enormous potential the mRNA technology disguises. Decades of research in various corners of medicine have allowed two biotech companies to develop COVID-19 vaccines in record time. Now, scientists want to take advantage of the surge of enthusiasm and investment to accelerate mRNA research in multiple areas of medicine. “It was a massive proof of principle,” says Preiss of the COVID vaccines.
“Who knows, one day we might have the ability to [make mRNA therapies] on a small scale at GP clinics depending on what the patient needs,” says Fox. “That’s real blue-sky thinking, but it is possible and quite exciting.”
But mRNA therapeutics – for heart disease and cancer treatment, for instance – might have a tougher road to the clinic than vaccines. These drugs face the challenges of targeting specific tissues and giving strong, long-lasting benefits without excessive side effects.
“We need to get these drugs to the right part of the body, at the right concentration,” says Leedman.
Tailoring an mRNA drug to a disease often means tweaking the structures of both the mRNA itself and the lipid nanoparticle that carries it through the body. Nanoparticle drug delivery research has an even longer history than the idea of mRNA therapy itself and has also regained momentum with the success of the COVID-19 vaccines.
Leedman sees the “delivery problem” as an opportunity rather than a challenge. “Who would have ever thought that medical research would be sexy?” he asks.
It turns out all that was needed was a global pandemic.
This post to mark World Cancer Day 2022 is an extract of an article on mRNA therapies that originally appeared in Issue 92 of Cosmos.
Years: 8, 9, 10, 11, 12
Login or Sign up for FREE to download a copy of the full teacher resource